本期封面
Tunable viscoelastic collagen/polyethylene glycol composite hydrogels modulate neural and tumor cell behavior in 3D microenvironments
Hexu Zhang, Ziyan Chen, Runxiang Yao, Yuyun Liang, Chaoyong He, Jing Yang, Houzhi Kang, Liyang Shi
本期文章共 10 篇,Editorial 1 篇,Research Article 3 篇,Review 4 篇,Commentary 2 篇。
Editorial
Advancing biomedical innovation through composite material strategies
Song Chen, Wei Xia*, Bin Li*
内容简介
This review summarizes recent advances in the spatiotemporal application of small molecules for fracture healing. It systematically discusses stage-specific signaling pathways involved in inflammation, repair, and remodeling, together with representative small-molecule modulators. In addition, emerging delivery strategies enabling sequential and stimuli-responsive release are highlighted. The review also addresses current challenges in pharmacokinetic control and clinical translation, providing perspectives for precision bone regeneration therapies.
DOI: doi.org/10.12336/bmt.25.00252
REVIEW
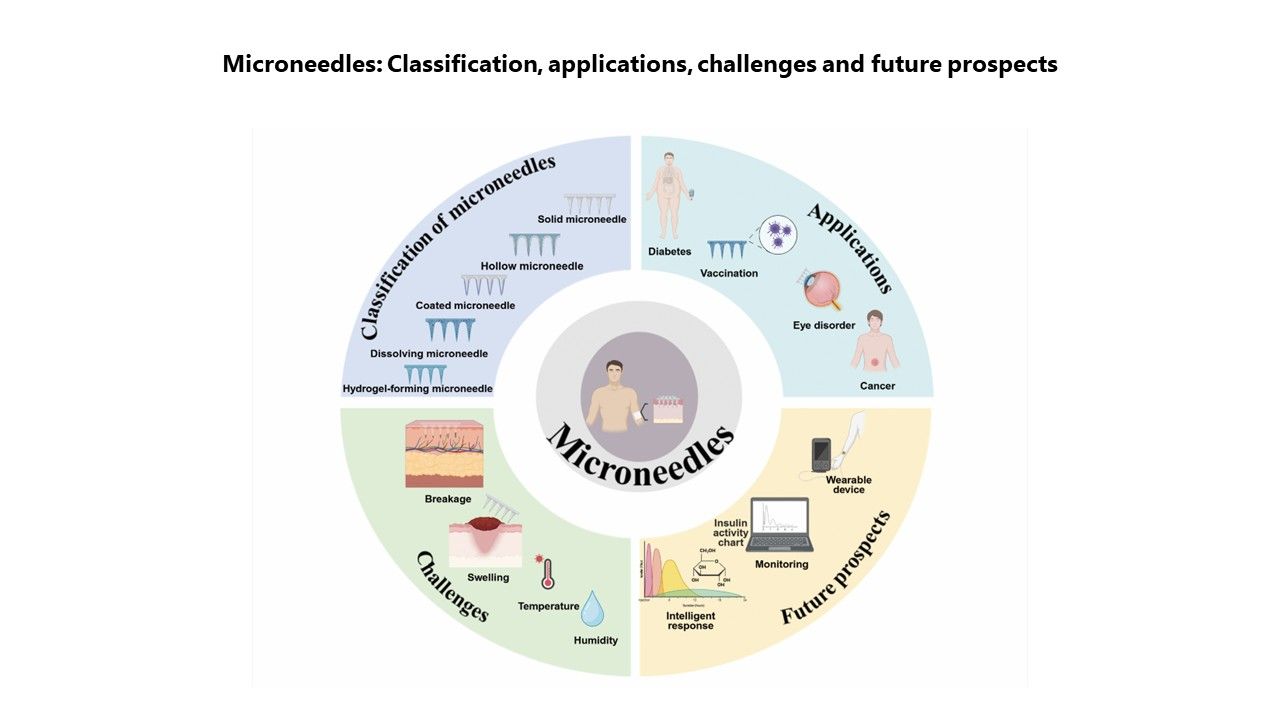
Microneedles in biomedicine: Innovations, challenges, and future prospects
Xinrui Li, Chi Zhang, Yuxin Zhang, Zhijing Liu, Jiaxin Li, Ying Meng, Peng Zhang*
内容简介
The effective delivery of therapeutic drugs is fundamental to modern medical practice. However, conventional administration methods, primarily oral and parenteral injection, exhibit numerous limitations, including the suboptimal bioavailability of macromolecules and challenges related to patient compliance. The advent of microneedle (MN) technology is reshaping strategies in the biomedical field, effectively overcoming the constraints of traditional drug delivery and diagnostic approaches. Research indicates that MNs can penetrate the stratum corneum to form transient microchannels, facilitating the transdermal delivery of therapeutic agents while bypassing gastrointestinal and hepatic barriers. This customizable and personalized drug delivery system holds significant potential for clinical application. Beyond drug delivery, MNs also have the capacity to transform healthcare models through real-time biomarker monitoring enabled by contact with interstitial fluid. This technology demonstrates considerable promise in managing chronic conditions such as diabetes, while also opening avenues for applications in vaccination, tissue regeneration, and cancer therapy. Recent innovations include the development of stimulus-responsive MNs for precision medicine and their integration with wearable devices to achieve closed-loop therapeutic diagnostics. Despite the substantial promise of this field, challenges remain regarding clinical translation, particularly in relation to biocompatibility, mechanical strength, and drug stability. This review outlines MN classifications, design principles, and applications, emphasizing their expanding role not only in healthcare but also in precision medicine, global health, and food safety. By overcoming current barriers and integrating emerging technologies, MNs have the potential to transform diagnostic and therapeutic paradigms, delivering scalable, patient-centered solutions to a broad range of biomedical challenges.
DOI: doi.org/10.12336/bmt.25.00122
REVIEW
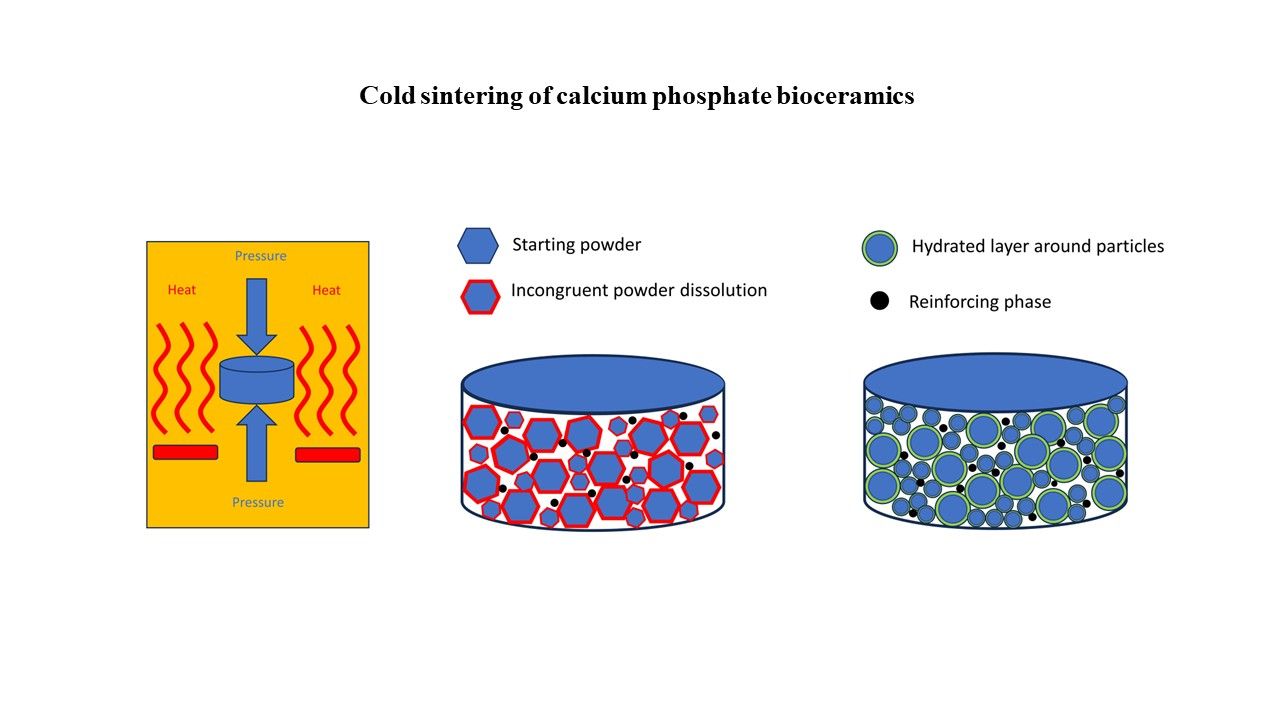
Cold-sintered bioceramics for medical applications: State of the art and further perspectives
Miodrag J. Lukic*, Denis Gebauer, Bin Li, Song Chen
内容简介
Cold sintering has recently emerged as a promising approach for preparing dense ceramic materials and composites at low temperatures. It relies on utilizing transient, typically externally introduced, liquid phases to accelerate material diffusion and densification under applied pressure. Cold-sintered bioceramics, especially those prepared at temperatures below 100°C, may open up numerous possibilities, not only in producing dense ceramics with refined microstructural properties and reduced time/energy costs, but also in developing multifunctional platforms containing bioactive compounds, therapeutics, growth factors, and signaling molecules for enhanced and targeted biological responses. Cold sintering in the presence of liquids inherently involves dissolution and nucleation, which become particularly intricate under applied pressures and elevated temperatures. Pseudo bio-mineralization, an auspicious approach for tailoring synthetic bone grafts toward targeted mechanics, may serve as a viable route for enhancing the densification mechanisms inherent to cold sintering. We have carefully analyzed the current state of the art in cold-sintered bioceramics and the results achieved, with a focus on the chemistry of the employed liquids and the corresponding changes upon sintering, the selection of transient phases, and mineral nucleation, while also addressing the potential for developing new biomaterials. Despite the widely accepted classical dissolution– precipitation strategy, no clear roadmap can yet be defined regarding the type and amount of liquid phase that should be applied, at least in the case of hydroxyapatite (HAp) densification–the most important representative of calcium phosphates. We strongly advocate the use of water as the transient liquid of choice in the cold sintering of HAp-based bioceramics, instead of strong acids/bases, and emphasize the importance of understanding the various processes and parameters that govern and connect solution chemistry to mineral nucleation. This understanding will enable the advancement of cold sintering protocols in a target-oriented manner, and we provide perspectives on future developments, including practical advice.
DOI: doi.org/10.12336/bmt.25.00157
REVIEW
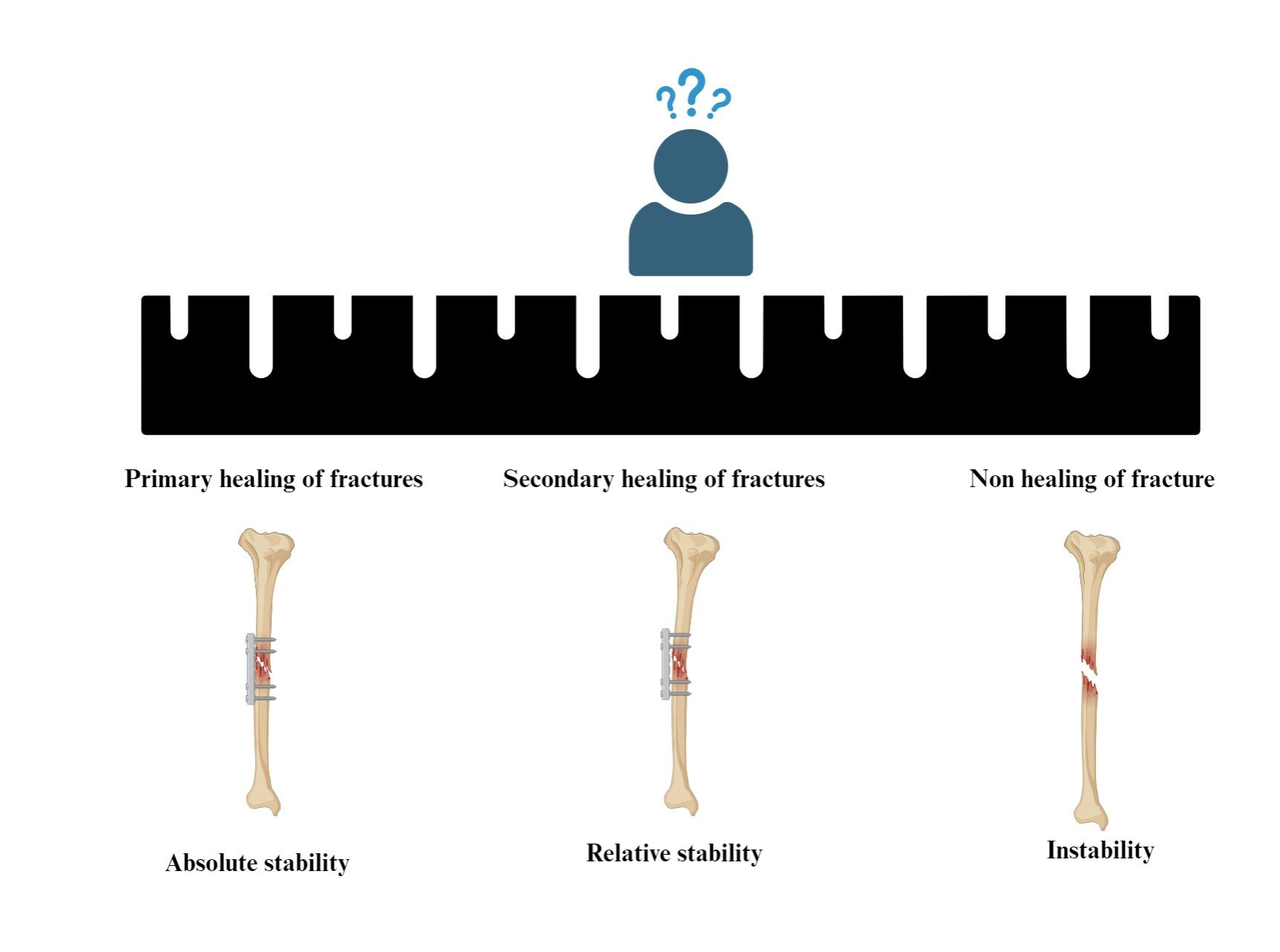
An exploration into the principles and theoretical progress of fracture treatment based on the mechanism of fracture healing
Haiming Ye#, Qi Yang#, Jiadi Hou, Xuan Zhang, Yien Zheng, Tiantian Qi*, Fei Yu*
内容简介
Fractures are a common category of diseases in the field of orthopaedics with a high incidence in archaeologically obtained bones. These diseases may occur in various human activities. In the context of technological advancement, the annual incidence of fractures is increasing due to traffic accidents, sports injuries, and ageing. Besides, the classification of fracture diseases is also changing, making them one of the main orthopaedic diseases that affect the quality of life of patients and national medical expenditure. There are some basic principles in the treatment of fractures, and the understanding of the causes, types, and pathogenesis of fractures is constantly improved with technological development. Hence, there are sustained efforts to explore fracture treatment methods and examine even widely popular concepts, such as Arbeitsgemeinschaft für Osteosynthesis (AO) and biological osteosynthesis (BO) principles. However, nonhealing fractures, fracture infections, and other treatment problems can still not be eliminated based on these concepts. In addition, some new perspectives on the treatment principles of fractures have been proposed by surgeons based on their clinical experience. In this paper, the latest research results on fracture healing are summarised, and our views and opinions on the application of AO or other new concepts in fracture treatment are also elucidated. During the investigation of the advantages and disadvantages of fracture treatment concepts, the shortcomings of current fracture treatment strategies or theories are also reviewed. These findings may provide clinicians with theoretical support for fracture treatment and inspire scholars to delve into fracture treatment principles.
DOI: doi.org/10.12336/bmt.24.00069
REVIEW
Spatiotemporal application of small molecules in fracture healing
Hairu Sui#, Zhonglin Wu#, Ziqi Xiong, Hui Zhang, Boon Chin Heng, Jing Zhou*
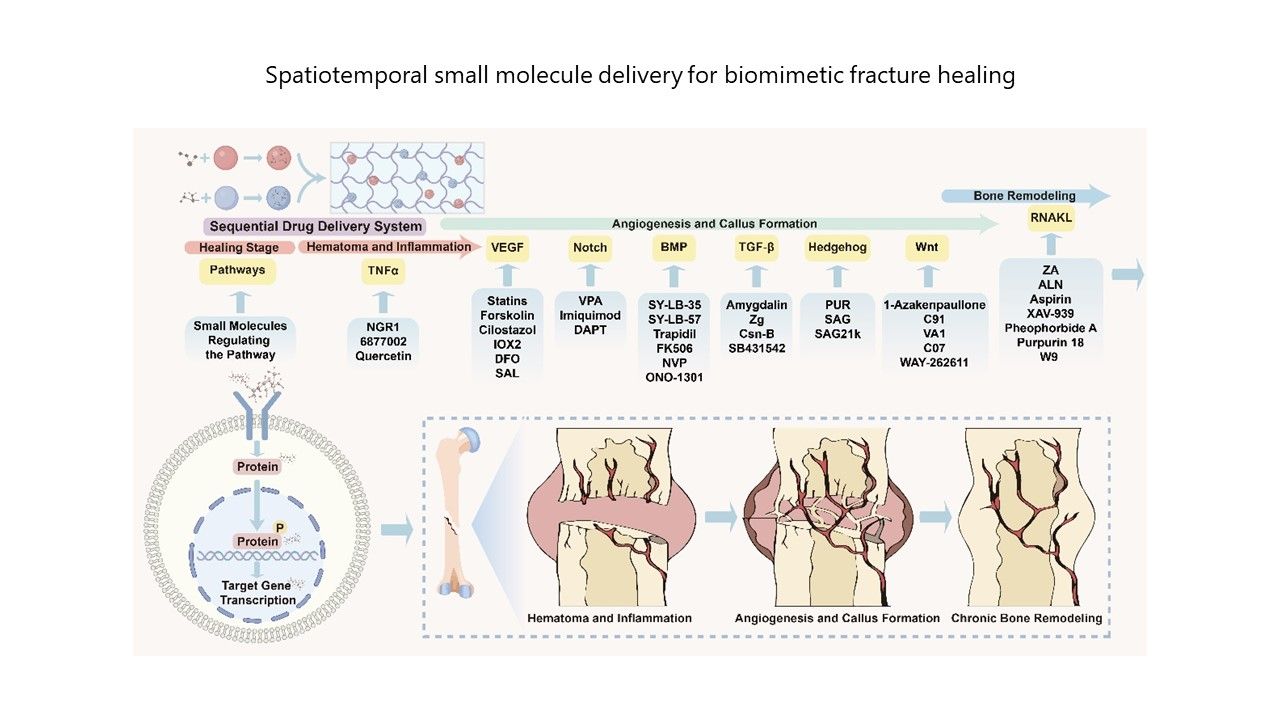
内容简介
Skeletal injuries and disorders are major causes of physical disability worldwide, posing an intractable clinical challenge. Within the field of regenerative medicine, researchers are continuously developing new therapeutic strategies to promote bone regeneration. Small molecules, defined as bioactive compounds with a molecular weight of <1,000 Da, have emerged as promising agents capable of precisely regulating intracellular signaling pathways to enhance bone regeneration. Their cost-effectiveness, superior membrane permeability, and minimal immunogenicity have positioned them at the forefront of both fundamental research and clinical applications. In recent years, advancements in artificial intelligence have accelerated the development and screening of small-molecule drugs, broadening their potential therapeutic applications. Furthermore, innovations in dynamic drug delivery systems have advanced the concept of spatial precision, enabling the controlled release of drug doses over time and achieving the spatiotemporal application of small molecules. These systems release specific small molecules in a sequence, synchronizing therapeutic interventions with the dynamic process of bone healing. Spatiotemporal delivery strategies, which effectively replicate the complex and highly ordered processes of bone healing, have the potential to reduce drug side effects and enhance healing efficacy. However, clinical translation remains hindered by insufficient spatiotemporal control and limited pharmacokinetic precision, challenges that this review explores in depth. We systematically examine stage-specific molecular targets of signaling pathways and their corresponding small molecule modulators. In addition, we discuss current approaches to spatiotemporal delivery strategies, such as stimuli-responsive delivery systems. Finally, we explore the status of clinical applications, the challenges encountered, and potential solutions regarding the spatiotemporal release strategy. We hope this review will contribute to the development of future spatiotemporal delivery strategies, ultimately improving outcomes for patients with impaired fracture healing.
DOI: doi.org/10.12336/bmt.24.00087
RESEARCH ARTICLE
Tunable viscoelastic collagen/polyethylene glycol composite hydrogels modulate neural and tumor cell behavior in 3D microenvironments
Hexu Zhang#, Ziyan Chen#, Runxiang Yao, Yuyun Liang, Chaoyong He, Jing Yang, Houzhi Kang, Liyang Shi*
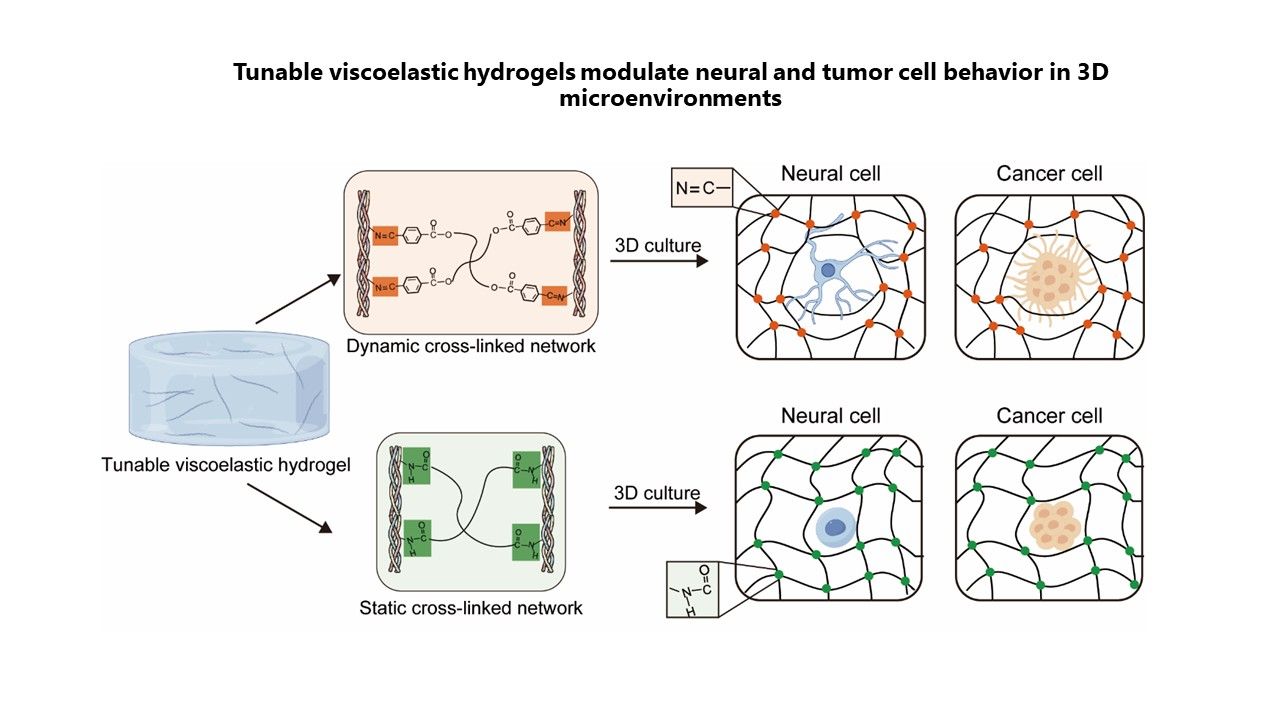
内容简介
Three-dimensional (3D) cell culture systems provide a more physiological environment than traditional two-dimensional cultures by better mimicking the complex interactions within the extracellular matrix (ECM). Among the key properties of the ECM, viscoelasticity is essential for regulating cell behaviors, such as proliferation, differentiation, and migration. However, many present 3D culture systems are complex and technically demanding, which limits their broad application. In this study, we developed two hydrogel systems with identical stiffness but distinct viscoelastic properties, designed to serve as ECM-based 3D culture platforms. These hydrogels were constructed through the cross-linking reaction between type I collagen and functionalized polyethylene glycol derivatives, resulting in either reversible (dynamic) or stable (static) network structures. This platform effectively simulated ECM-like mechanical cues, enabling the investigation of viscoelastic effects on both neural and cancer cell responses. Our results demonstrated that dynamic hydrogels, characterized by rapid stress relaxation, enhanced PC12 cell elongation, promoted neural stem cell differentiation, and significantly facilitated the invasiveness and tumorigenic capacity of DU145 cells in vitro and in vivo. These findings highlight the critical importance of matrix viscoelasticity in modulating cell behavior and underscore the potential of this hydrogel-based system as a versatile and accessible tool for applications in neural tissue engineering, cancer research, and mechanobiology.
DOI: doi.org/10.12336/bmt.25.00096
RESEARCH ARTICLE
Recombinant human bone morphogenetic protein-2–engineered piezoplatform synergistically promotes bone regeneration through bone morphogenetic protein receptor activation
Lijie Mao, Dong Zhang, Zehao Shen, Xinqing Wang, Chen Lai, Fangping Chen*, Changsheng Liu*
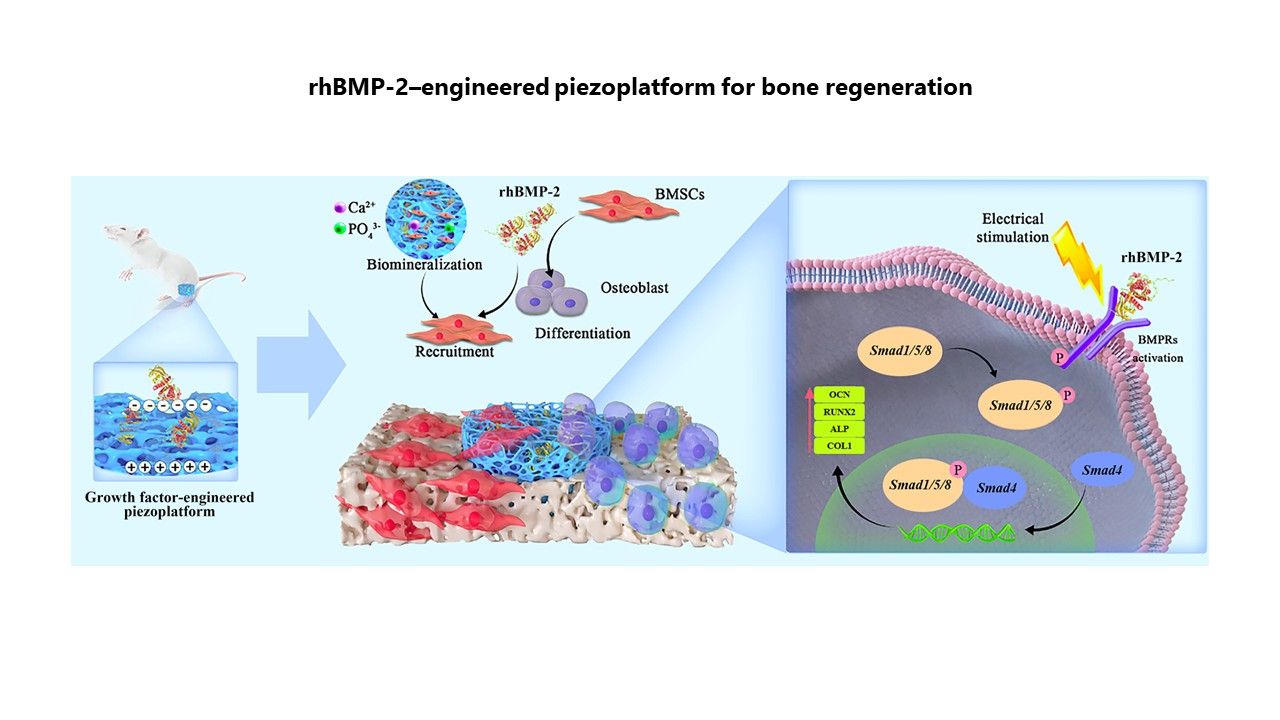
内容简介
Bone morphogenetic protein-2 (BMP-2) is a potent cytokine that promotes bone formation in orthopedic procedures. However, the delivery of recombinant human BMP-2 (rhBMP-2) with sustained release kinetics, while maximizing osteogenic potential, remains a challenge. In this study, we constructed a novel rhBMP-2–engineered piezoplatform for sustained release of rhBMP-2 and synergistic enhancement of osteoinductive activity. The piezoelectric signals are capable of initiating rapid biomineralization and promoting the early adhesion, proliferation, and osteogenic differentiation of bone marrow stromal cells (BMSCs), as well as enabling efficient immobilization and sustained release of rhBMP-2 through electrostatic interactions. Notably, piezoelectric stimulation synergizing with rhBMP-2 enhances osteogenesis-related protein production. This is achieved by amplifying the expression of BMP-2 receptors (Bmpr1a and Bmpr2) in BMSCs by approximately three-fold, which in turn reinforces the regenerative capacity of rhBMP-2. The rat femur defect model further confirms the osteogenic efficacy of the rhBMP-2–engineered piezoplatform. These findings are expected to advance the development of biopiezoelectric implants incorporating growth factor therapy for tissue engineering.
DOI: doi.org/10.12336/bmt.25.00019
RESEARCH ARTICLE
PBVHx-based microspheres for controlled BMP2 release and enhanced bone regeneration in a disuse osteoporosis mouse model
Kewen Zhang#, Yanwen Zhou#, Daixu Wei*, Airong Qian*, Xiao Lin*
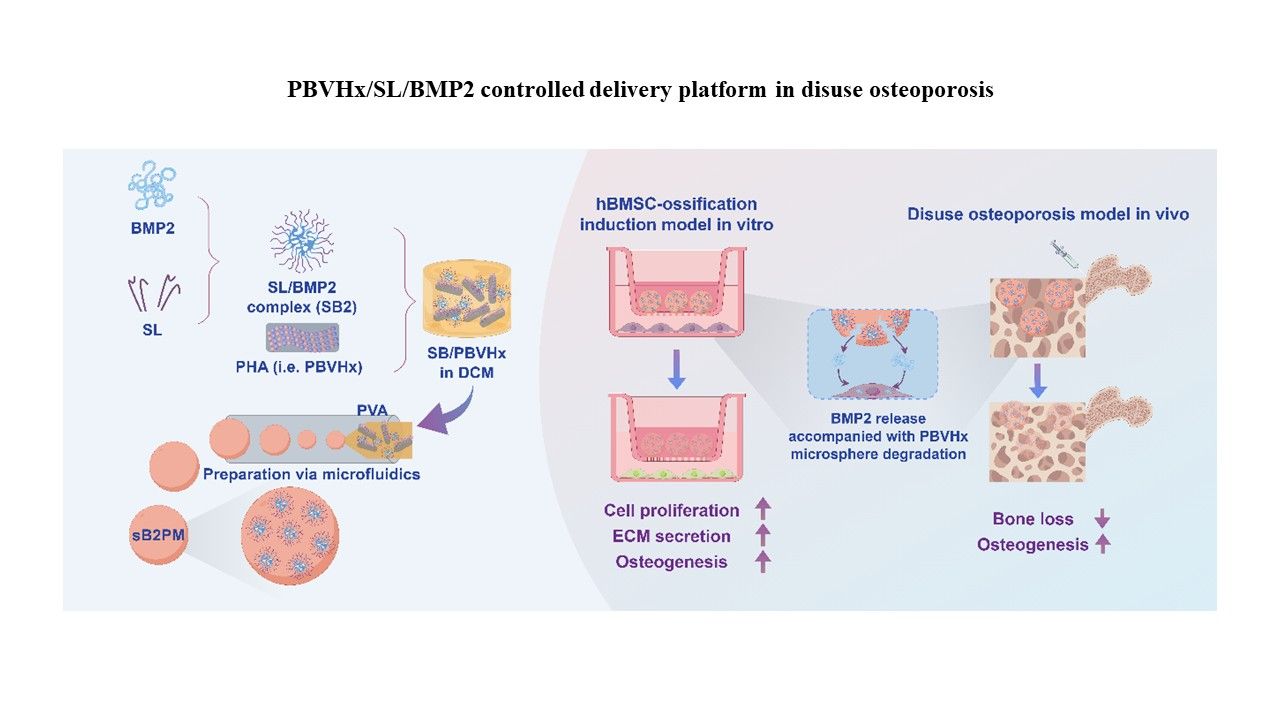
内容简介
Addressing bone defects caused by degenerative diseases, trauma, and cancer through bone tissue engineering remains a significant global health challenge. The osteoinductive properties of bone morphogenetic protein-2 (BMP2) have become a key therapeutic strategy in bone regeneration. However, the development of biodegradable composites that ensure biocompatibility, stability, efficient BMP2 loading, and controlled release remains unresolved. In this study, we designed PBVHx/soy lecithin (SL)/BMP2 controlled-release microspheres (sB2PM) based on the biodegradable material poly(3-hydroxybutyrate-co-3-hydroxyvalerate-co-3-hydroxyhexanoate) (PBVHx), incorporating SL to enable sustained BMP2 delivery and enhance capture. sB2PM microspheres exhibited uniform size (approximately 5 μm) and high BMP2 encapsulation efficiency (80.29%) compared to pure PBVHx-based microspheres (pPM). Due to PBVHx’s biodegradability, BMP2 release was primarily degradation-driven, resulting in a controlled biphasic release profile. sB2PM achieved 62.79% cumulative BMP2 release over four weeks and continued to release BMP2 sustainably thereafter. Co-culturing sB2PM microspheres with human bone marrow-derived mesenchymal stem cells (hBMSCs) in a Transwell system showed enhanced cell proliferation, biocompatibility, and collagen secretion. Compared with pPM and B2PM, sB2PM significantly promoted osteogenic differentiation, increased alkaline phosphatase (ALP) activity, and upregulated osteogenic gene expression in hBMSCs, outperforming commercial hydroxyapatite microspheres. In a mouse hindlimb unloading osteoporosis model, micro computed tomography and histological evaluations confirmed that injectable sB2PM microspheres significantly enhanced bone regeneration, collagen secretion, and ALP and runt-related transcription factor 2 protein expression. This study highlights the potential of sB2PM microspheres with controlled BMP2 release for future bone regeneration therapies.
DOI: doi.org/10.12336/bmt.25.00072
COMMENTARY
Integrating nanomedicine and immunotherapy: Bacterial membrane–derived vesicle-encapsulated prodrug assemblies for chronic infections
Xinnan Zhong#, Jiaqi Chen#, Yijun Li, Zilin Zhou, Jiyao Li, Jun Luo*, Jiaojiao Yang*
内容简介
This commentary highlights a bio-inspired nanomedicine strategy that integrates antibiotic delivery with immunomodulation to address chronic bacterial infections. By encapsulating self-assembled antibiotic prodrugs within bacterial membrane–derived vesicles, the platform enables efficient penetration of biofilms, enhanced intracellular bacterial clearance, and reprogramming of innate and adaptive immune responses. This dual chemical–immunological approach offers a promising direction for overcoming immune evasion and recurrence in persistent infections.
DOI: doi.org/10.12336/bmt.25.00153
COMMENTARY
The advancement of composite materials in future biomedical technologies
Wei Xia*, Song Chen, Bin Li
内容简介
This commentary discusses the growing importance of composite materials in next-generation biomedical technologies. By integrating ceramics, polymers, metals, and functional additives, composites enable simultaneous optimization of mechanical performance, biological integration, and multifunctionality. The article highlights key design strategies, emerging applications in implants, tissue engineering, and drug delivery, and outlines major translational challenges related to predictability, manufacturing, and regulation.
DOI: doi.org/10.12336/bmt.25.00237
本期封底
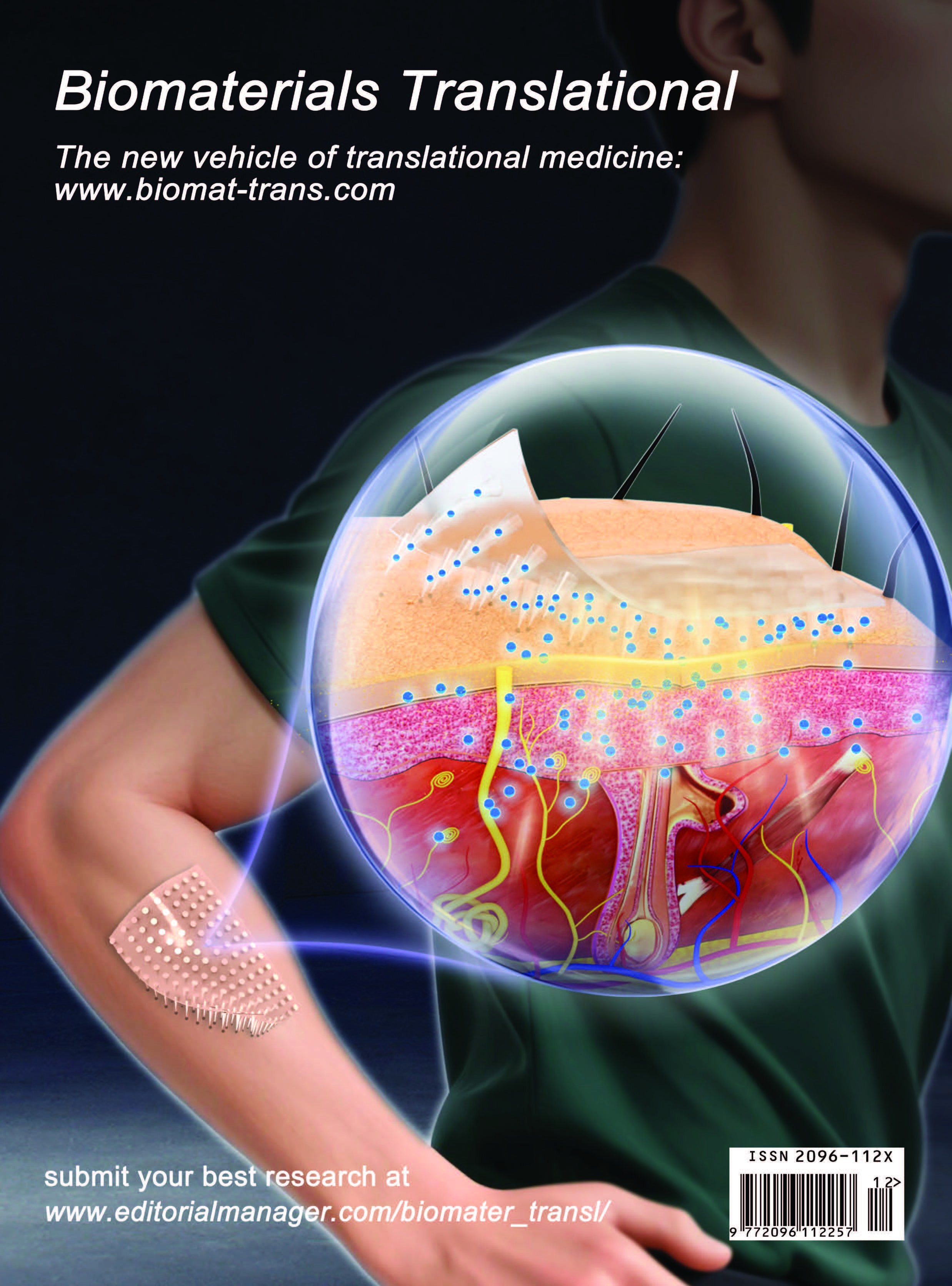
Microneedles in biomedicine: Innovations, challenges, and future prospects
Xinrui Li, Chi Zhang, Yuxin Zhang, Zhijing Liu, Jiaxin Li, Ying Meng, Peng Zhang


